Disculpa, pero esta entrada está disponible sólo en English.
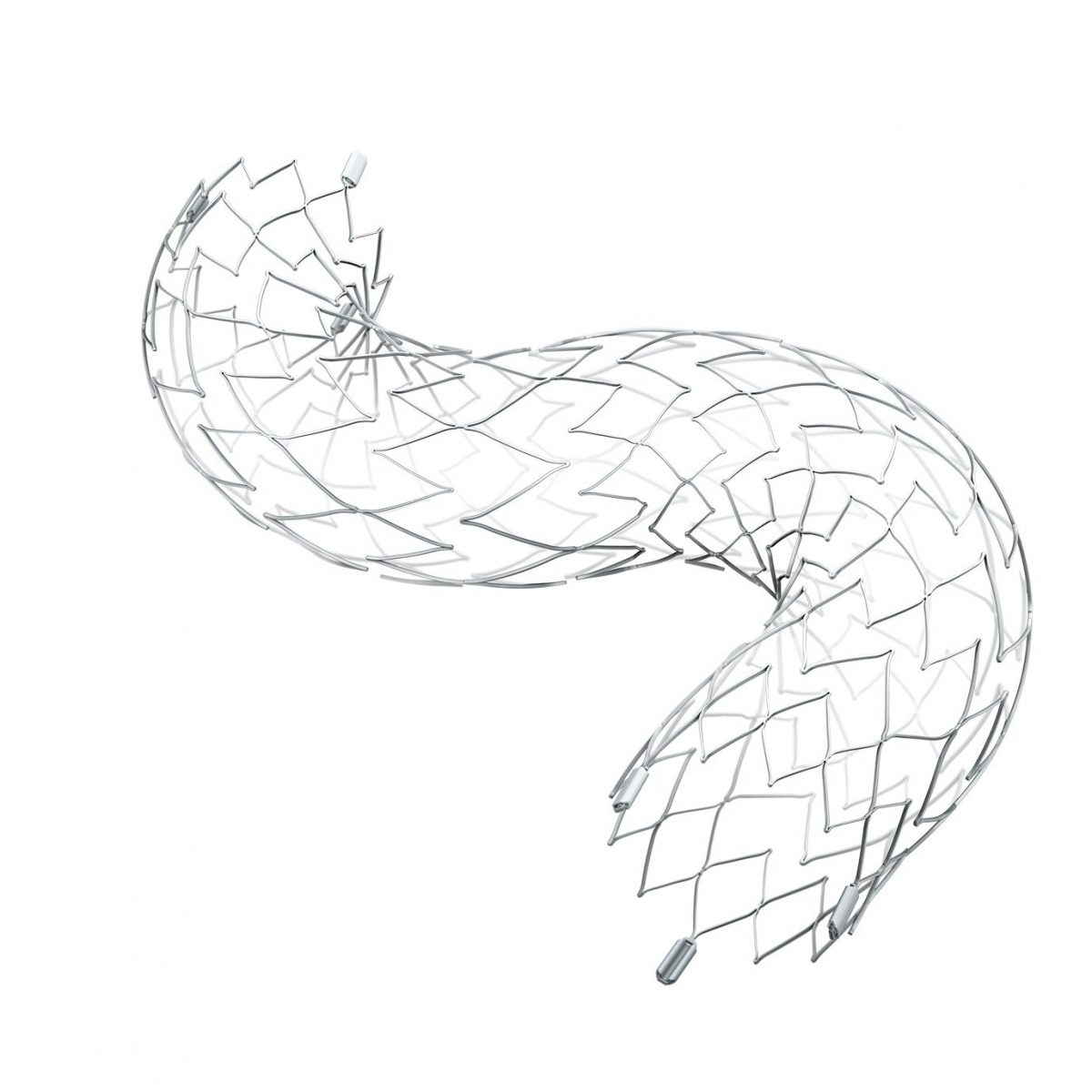
(English) Stryker’s Neuroform Atlas® Stent System granted an expanded indication, providing a new option for patients with aneurysms in the back of the brain
(English)
KALAMAZOO, Michigan, USA, Aug. 3, 2020 /PRNewswire/ — Stryker announced today that it has received U.S. Food and Drug Administration (FDA) approval for an expanded indication of its Neuroform Atlas Stent System, becoming the first and only adjunctive stent approved for use in the posterior (back of the brain) circulation. Aneurysms in the posterior circulation rupture more frequently and are generally more difficult to treat. With the approval of the Neuroform Atlas adjunctive stent for…